Mounjaro vs Wegovy — The Complete UK Comparison 2026
Mounjaro vs Wegovy – The Complete UK Comparison 2026
Written on 7th May 2026, by Dr Weightmans
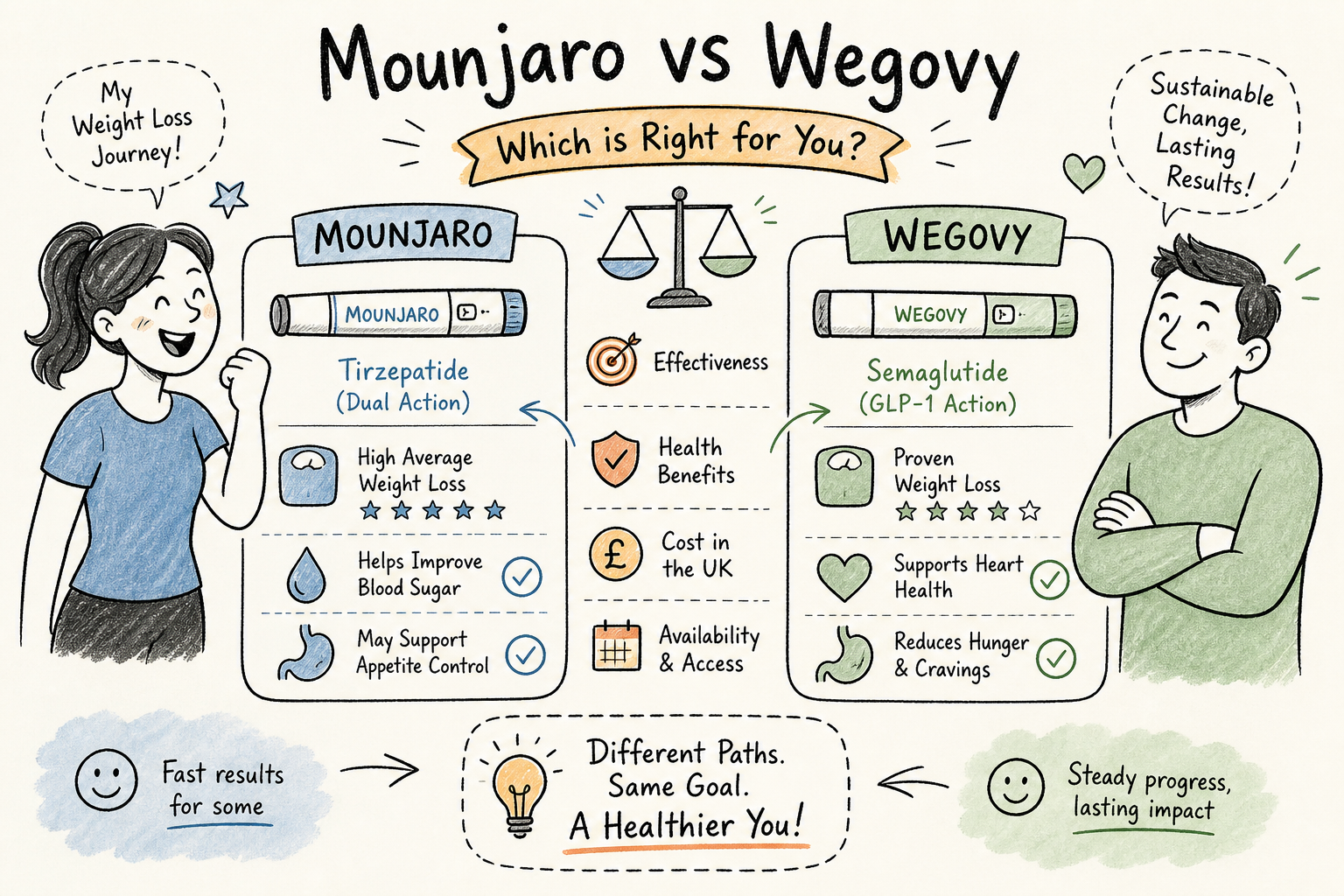
Mounjaro vs Wegovy: both are weekly injections rooted in GLP-1 science, both are prescription-only in the UK, and both are clinically proven to produce meaningful weight loss. So are they actually that different, or is this just pharmaceutical branding at work?
The short answer: they are genuinely different, and the clinical trial data makes the gap impossible to ignore. This comparison draws on published RCT results, UK regulatory approvals, and supporting evidence across five areas: mechanism, efficacy, dosing, side effects, and UK access in 2026. It covers tirzepatide vs semaglutide from first principles so you can walk into a clinical conversation knowing exactly what to ask.
This is not an attempt to sell you one drug over the other. It is grounded information to help you make sense of a real clinical decision.
How Mounjaro and Wegovy work differently
Semaglutide’s single-hormone approach
Wegovy contains semaglutide, a GLP-1 receptor agonist. GLP-1 (glucagon-like peptide-1) is a gut hormone released naturally after eating. It signals fullness to the brain, slows gastric emptying, and suppresses glucagon to reduce blood sugar. Semaglutide mimics this hormone at a sustained level throughout the week. It is a single-receptor agonist: well-understood and effective at producing meaningful appetite suppression in most people who use it.
Tirzepatide’s dual-hormone advantage
Mounjaro contains tirzepatide, which activates two receptors simultaneously: GLP-1 and GIP (glucose-dependent insulinotropic polypeptide). The GIP pathway adds a second mechanism for appetite suppression and improves insulin sensitivity through a distinct biological route. What makes this distinct is that tirzepatide was engineered by embedding GLP-1 activity into the GIP molecular sequence, and it requires lower GLP-1 receptor engagement than semaglutide to achieve equivalent glycaemic control. That suggests tirzepatide is doing more metabolic work per unit of GLP-1 activity, not just doubling up on the same signal.
What the difference means in practice
This dual mechanism is not theoretical. It produces measurable differences in triglyceride reduction, insulin sensitivity, and total body weight loss that go beyond what weight reduction alone would explain. The metabolic benefits of tirzepatide appear partly independent of its effect on weight, which matters for patients with insulin resistance or type 2 diabetes. That mechanism gap may help explain why the efficacy data diverges as sharply as it does.
Mounjaro vs Wegovy: what the clinical trials actually show
STEP and SURMOUNT: the individual trial results
The STEP trials for semaglutide 2.4mg reported mean body weight loss of 14.9 to 17.4% at 68 weeks. The SURMOUNT trials for tirzepatide 15mg reported 20 to 22.5% at 72 weeks. These were separate trials with different patient populations, which is a legitimate caveat. The direction is consistent across every analysis, though it is worth noting that pooled data show substantial heterogeneity across individual studies; even so, the magnitude of difference is large enough that population variation alone is unlikely to account for it. For primary trial sources, see the STEP trial publications on PubMed.
The head-to-head result from SURMOUNT-5
The SURMOUNT-5 trial, published in the New England Journal of Medicine, compared tirzepatide directly against semaglutide 2.4mg in 751 adults without diabetes over 72 weeks. Tirzepatide produced 20.2% mean body weight loss. Semaglutide produced 13.7%. That is a 47% relative advantage for tirzepatide, statistically significant across every secondary endpoint. You can read the full SURMOUNT‑5 publication for trial details in the published paper.
Waist circumference reductions were 18.4cm versus 13.0cm. And 31.6% of tirzepatide participants achieved at least 25% body weight loss, compared to 16.1% on semaglutide. These are not marginal statistical differences. They represent a clinically meaningful gap in outcomes. Independent news coverage also highlighted the head-to-head result and its implications for clinical practice (coverage of the head‑to‑head result).
What 20% versus 13.7% actually means for a real person
For someone starting at 100kg, the difference between these averages is roughly 6.5kg more lost on Mounjaro at the same treatment duration. At 120kg, that gap widens to around 8kg. Individual variation is real: some patients lose more on semaglutide than the tirzepatide average. But the population-level advantage for tirzepatide is consistent across trials and meta-analyses, with pooled data showing a mean difference of around 4.6 to 5.0% additional body weight in favour of tirzepatide at higher doses.
Mounjaro vs Wegovy: dosing schedules and what treatment looks like week to week
Wegovy’s 16-week titration path
Wegovy follows a four-step titration: 0.25mg in weeks one to four, 0.5mg in weeks five to eight, 1.0mg in weeks nine to twelve, 1.7mg in weeks thirteen to sixteen, then 2.4mg as the maintenance dose from week seventeen. Most patients reach the therapeutic maintenance dose after approximately sixteen weeks. Some stay at a lower dose if side effects are significant during titration, and that is a clinical decision made with a prescriber, not a self-directed one.
Mounjaro’s 20-week titration path
Mounjaro uses six dose steps from 2.5mg to 15mg, increasing in 2.5mg increments with a minimum of four weeks between each step. The starting dose of 2.5mg is for adjustment only. It is not yet at a therapeutic level for weight loss. Full titration to the maximum 15mg dose takes at least twenty weeks. In practice, many patients find an effective maintenance dose before reaching 15mg and stay there rather than pushing to the maximum.
Flexibility and what this means before you start
Neither drug is a rapid ramp-up. Both require patience during titration, and the headline weight loss numbers from clinical trials reflect the full treatment duration, not the first few weeks. If you miss doses or need to stop and restart for any reason, re-titration guidance from a prescriber is essential. Starting lower and moving up slowly is not a failure of the treatment; it is how both drugs are designed to work.
Side effects: what most people actually feel
The shared side effects: mostly GI, mostly manageable
When comparing Mounjaro vs Wegovy, you may want to consider their corresponding side effect profiles. Both drugs slow gastric emptying, which means nausea, diarrhoea, and vomiting are the most common complaints on either. Tirzepatide trials report nausea in 30 to 40% of participants. Roughly three-quarters of Wegovy users in trials reported some GI disturbance. For most people, these effects are worst during dose increases and improve as the body adapts. Injection-site reactions and reduced appetite are also common to both and are, in many respects, the intended effect.
Where the profiles differ
In several trials, tirzepatide was associated with fewer severe GI events compared to semaglutide, though discontinuation rates varied across studies and the picture is not uniform. Mounjaro carries a specific caution against use in patients with gastroparesis. Semaglutide has a more documented association with psychiatric adverse events, including depression and anxiety, based on post-market safety reporting. Neither signal should be overstated, but both are worth raising with a prescriber, particularly if you have a relevant history.
Serious adverse events: rare but documented
Both medications carry a label warning for thyroid C-cell tumour risk observed in rodent studies and are contraindicated in patients with a personal or family history of medullary thyroid carcinoma or MEN 2, in line with their respective UK Summary of Product Characteristics. Pancreatitis and kidney-related complications have been linked to both drugs in real-world data. UK prescribers screen for all of these contraindications before issuing a prescription, which is one of the key reasons these are prescription-only treatments and not available over the counter.
UK cost, access and eligibility in 2026
NHS access: the eligibility criteria and the wait
On the NHS in 2026, Mounjaro is available for adults with a BMI of 40 or above with at least four weight-related comorbidities, with eligibility expanding to BMI 35 to 39.9 with four or more conditions from June 2026. Wegovy’s NHS eligibility is set at BMI 35 or above, or BMI 30 to 34.9 with at least one comorbidity, accessed through specialist weight management services. The reality of NHS access is that waiting lists for these specialist services range from three to twelve months in most areas, with some regions reporting waits of over two years. Being eligible on paper does not mean starting treatment soon, see the NHS backlog data for more on wait times and regional variation.
Private prescription: what each drug costs in 2026
For patients who do not want to wait, private prescriptions are available for both medications. In 2026, Mounjaro typically costs between £130 and £375 per month depending on dose, with starter doses at the lower end and maintenance doses at the higher end. Wegovy ranges from approximately £124 to £324 per month across equivalent stages. Prices vary by provider and have shifted notably since late 2025, particularly for higher Mounjaro doses. The private route mirrors NHS eligibility thresholds (BMI 27 or above with relevant comorbidities, or BMI 30 or above) but removes the waiting list entirely. Dr Weightmans is among the most cost effective, if not the cheapest provider throughout the weight loss journey in the UK.
Getting the right guidance before you choose
Comparing Mounjaro and Wegovy is a useful starting point, but the final decision should involve a UK-registered prescriber who can review your full clinical picture. Dr Weightmans is a GPhC-registered online pharmacy and prescribing service specialising in weight management. Online consultations are reviewed by UK-registered prescribers who assess clinical eligibility, review your health profile, and help identify which medication fits your goals and medical history. If you are already using Mounjaro or Wegovy, and are thinking of changing to the other, you can refer to the helpful switching guide on the Dr Weightmans website.
Which medication is likely right for you
When Mounjaro is the stronger clinical choice
Tirzepatide’s dual mechanism and superior average weight loss data make it the stronger option for patients with a higher BMI who need substantial weight reduction, those with insulin resistance or type 2 diabetes where the GIP pathway provides additional metabolic benefit, and patients who have already tried semaglutide and found the results insufficient. A 20%-plus average weight loss at 72 weeks is a meaningful clinical outcome for anyone managing serious weight-related health conditions.
When Wegovy is worth considering
Wegovy remains a clinically proven, effective treatment. It suits patients with a lower BMI or a more modest weight loss target where tirzepatide’s greater potency is not required. It has a longer real-world safety record in the UK market, and for patients with established cardiovascular disease, the SELECT trial evidence supporting semaglutide for cardiometabolic outcomes is well-established. For some patients, the side-effect profile may also prove more predictable given the longer post-market history.
The next step is a proper consultation, not a self-decision
Both Mounjaro and Wegovy are prescription-only in the UK for good reason. The best answer to “which drug?” comes from a UK-registered prescriber who can review your BMI, medical history, comorbidities, and treatment goals in full. A comparison article can frame the decision; a clinical consultation makes it. If you are ready to find out which medication fits your situation, that is exactly what a free consultation is designed to address.
The bottom line
When weighing Mounjaro vs Wegovy, both represent genuine advances in medically supported weight loss, backed by strong trial evidence and meaningful real-world results. The data consistently shows tirzepatide producing greater average weight loss, and the SURMOUNT-5 head-to-head result is difficult to dismiss. But greater on average does not automatically mean right for you. The individual clinical picture matters more than the headline numbers.
Dr Weightmans provides access to both medications through a fully regulated, GPhC-registered prescribing service, with online consultations reviewed by UK-registered prescribers for anyone considering either drug. If you are comparing your options and want a clinical assessment rather than a self-diagnosis, that is exactly what the consultation is for. Start there.







